 This number is representative of momentum or how fast the object is travelling. We measure the wave pattern between two peaks and get a number. We can determine the momentum of a quantum object with a simple calculation. The uncertainty principle is applicable to many more particles, but we’ll use these as examples. Many of us will have heard of radio waves or light waves for example, but sound and light can also be described as phonons (particles of sound) and photons (particles of light). This means they are both a wave and a particle at the same time. One of the key things to try and understand in this topic is that quantum objects have a property called ‘wave particle duality’. A typical wave Duality, two things at the same time We’re all pretty familiar with waves, at least the ones we see on water anyway so lets visualise one of those and draw a representation it below. The little triangle is the Greek letter ‘Delta’ and in physics we use it to represent a change. It saves us writing 0.0000000000 and so on over and over again, making the equation even more daunting looking! This is a very small unit of measurement close to zero. The ‘h’ term is something called ‘Planck’s Constant’. It looks and sounds complicated but we only need to understand some of it to get the gist.ĭelta X is uncertainty in position, Delta P is uncertainty in momentum. You would read this equation as follows: Delta X multiplied by Delta P is greater than or equal to h over 4 pi. Let’s look at it anyway, its does us good to gain more exposure to these equations as we try to build our knowledge and understanding of these subjects. We don’t have to, to be able to gain a high level understanding of it. It doesn’t matter too much that we don’t fully understand all the terms precisely. Heisenberg’s uncertainty principle is represented by an equation, which I’ve written below.  
Things this small behave extremely strangely indeed and we find it incredibly difficult to make accurate predictions about them, most notably their position or ‘place’ and their momentum or ‘speed’. We would write that size scientifically like this 1× 10 − 9 m. Typically an object that can only be described by quantum mechanics is 1 nano metre in size or smaller, that’s 0.000000001 metres or one billionth of a metre. We call these small things ‘quantum objects’.Ī quantum object is any object that cannot be defined by standard physical laws and must be defined by the laws of quantum mechanics. The uncertainty principle is a scientific fact of the natural world, albeit the natural world of incredibly small things. 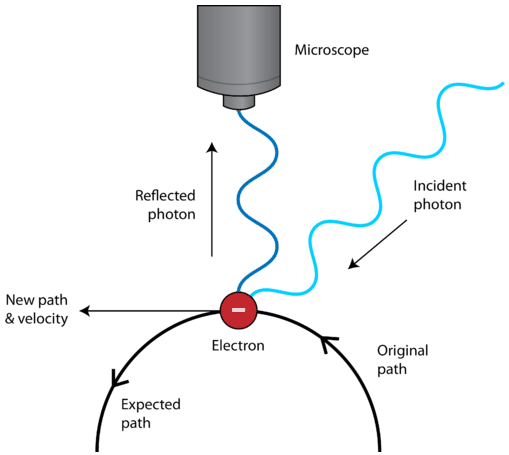
Let’s see if we can understand both the principle itself and why Walter might have cleverly chosen it to represent his character. It’s name and underlying message were introduced firmly into pop culture more recently in the TV show ‘Breaking Bad’ where lead protagonist Walter White chose the name ‘Heisenberg’ as his pseudonym. It was first postulated by German physicist Werner Heisenberg in the late 1920’s. In this post we are going to examine and try to understand one of the more well established aspects of particle physics, the Heisenberg Uncertainty Principle. Reading Time – 8 minutes, Difficulty Level 4/5 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
